Home
An Oral and Non-toxic Cancer Treatment Option
Have you, a family member, or a friend been affected by cancer? Have you seen the effects of traditional chemotherapy medication treatments? Patients having to go to a cancer treatment center and sit through hours of an intravenous medication treatment? Having to endure the nausea, weight loss, hair loss, and lethargy that can accompany these treatments? CZ48 is a medication that is delivered via capsule and is very low to no toxicity that; consequently, allows cancer patients to avoid the negative side effects of traditional chemotherapy. Would you like to invest in a company that is developing non-toxic, orally delivered cancer medications?
Introducing Cao Pharmaceuticals Inc.
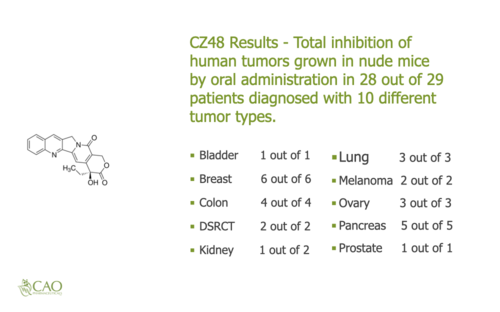
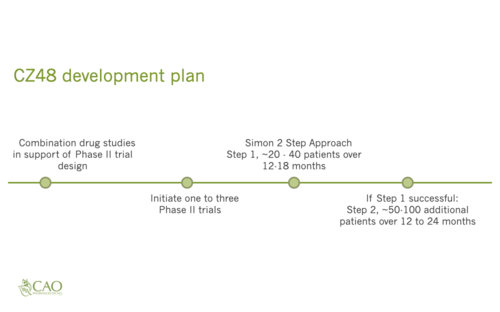
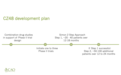
Cao Pharmaceuticals Inc. was founded by one of the leading camptothecin (cpt) researchers in the world. Dr. Cao and Cao Pharmaceuticals Inc personnel have successfully developed and licensed cpt medications. CZ48 is a cpt derivative that is delivered orally via capsule. During the recently completed CZ48 Phase 1 Food and Drug Administration (FDA) human trial determining dosage, CZ48 proved to have very low to no toxicity. During laboratory trials conducted implanting human cancers into nude mice and administering CZ48 orally to those nude mice, CZ48 totally inhibited 28 out of 29 of the human tumors for ten different types of cancer including Bladder, Breast, Colon, DSRCT, Kidney, Lung, Melanoma, Ovary, Pancreas, and Prostate. These are very promising results. The next step is to study the efficacy, how well CZ48 works in people, in the Phase 2 FDA human trial.
Pharmaceuticals Business Model
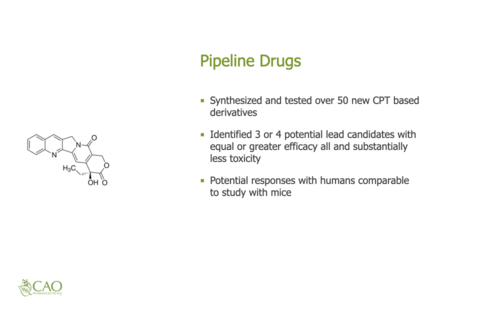
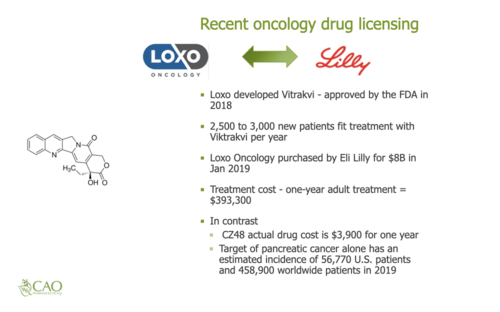
Cao Pharmaceuticals is a small research and development (R&D) company. Typically, small pharmaceutical companies develop new medications and license these medications to larger companies. These licenses are typically one-time, upfront payments per type of cancer. For example, a one-time fee for a license for Bladder cancer and another fee for Breast cancer. This licensing can occur prior to FDA approval. Secondly, there are annual royalties that are a percentage of annual sales that can range from single digits up to 25% that are ongoing annual payments. The sales royalties do not occur until after FDA approval. Thirdly, there are other medications in the development pipeline that could later be licensed and earn annual royalties. Fourthly, there is company ownership that provides ownership of the pipeline medications and company stock that could later appreciate. The structure of the pharmaceuticals business provides potential for both short-term and long-term investment returns.
Use of Investment Funds
The investment funds will be used as working capital to fund the Phase 2 FDA trial, paying trial expenses for third party conducted patient treatment and evaluation, laboratory rental, utilities, expenses, and ongoing pipeline medication development as well as Cao Pharmaceuticals personnel salaries.
Download the Silicon Prairie Online Mobile App to scan the QR Code for this page:
![]()